Germany, the biggest EU economy, is officially opening up to digital health innovation. From September 2020, DVG means digital health applications that get approved by BfArM (the German regulator) can be prescribed by doctors in Germany. Check our explanation of how your digital health application can get fast-track approved. In this blog post, we will answer the most common questions about this new German legislation.
The change Digital Care Act (DVG) is bringing to Digitale Gesundheitsanwendungen (DiGA)
Digital health applications (also known in German as Digitale Gesundheitsanwendungen, DiGA) become like "digital helpers" in the hands of the patient. Digital health apps from now will help doctors to collect data and help to make disease treatment more effective.

What is a DiGA?
For DVG, a DiGA (digital health application) is quite narrowly defined:
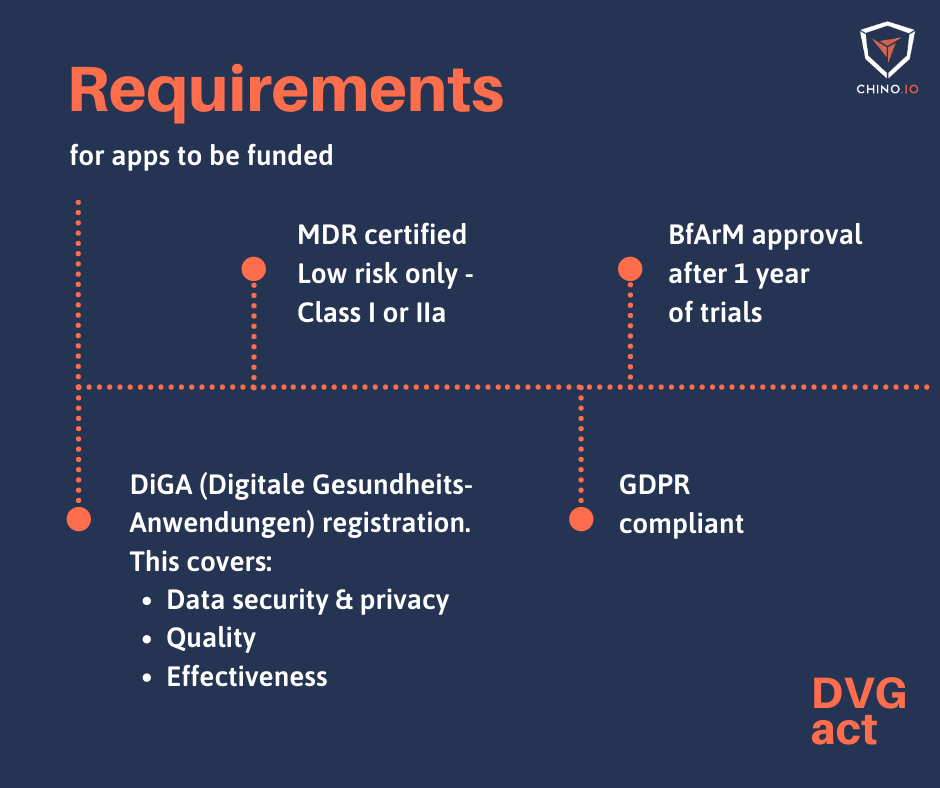
- Medical device of risk class I or IIa (according to MDR or, during the transitional period, according to MDD)
- The main function of the DiGA is based on digital technologies
- The main medical purpose is achieved through the main digital function
- The DiGA supports the detection, monitoring, treatment, or relief of diseases or the detection, treatment, relief, or compensation of injuries or disabilities
- The DiGA is shared between the patient or the service provider and patient
What is excluded from this?
Some digital health applications are excluded. For instance, any application with a higher-risk category cannot receive fast-track approval. Equally, if your application can’t receive an MDD or MDR certification, it can’t be included. Furthermore, some apps are already able to be reimbursed through other routes. This includes telemedicine apps that perform remote monitoring. Most significantly, apps that are just used by a doctor to diagnose or treat a patient are excluded (since they count as practice equipment). To count as a DiGA, it must be an app prescribed by the doctor that the patient uses themselves.

What are the benefits of the DVG?
The Digital Care Act (DVG) is a German law that came into force on December 19, 2019. DVG means that around 73 million insured persons in statutory health insurance are now entitled to care involving digital health applications (DiGAs). After digital health apps become approved, they can be prescribed by doctors and psychotherapists and reimbursed by the health insurance company.
How does your DiGA get approved?
Your DiGA has to go through a verification procedure at BfArM, the Federal Institute for Drugs and Medical Devices of Germany (in German Das Bundesinstitut für Arzneimittel und Medizinprodukte).
What is the directory of reimbursable digital health applications, DiGA-Verzeichnis?
After getting approved by BfArM, your app will be listed in a new directory of reimbursable digital health applications (DiGA-Verzeichnis). This directory summarises essential information about the available DiGAs for doctors, psychotherapists, and patients. This ensures extensive transparency, as all approved DiGAs will be listed there.
This is defined in the supplementary ordinance of the Federal Ministry of Health (BMG), the Digital Health Applications Regulation (DiGAV).
What is the BfArM assessment procedure?
The procedure is called the "Fast Track" because it takes around three months to get approved.
The goal is to assess whether your application meets the general requirements set out for approval. These cover a wide range from data protection to user-friendliness. In addition, there’s a need to prove the so-called “positive care effects” of the DiGA. However, BfArM gives you up to 12 months from approval to do this.
How can Chino.io help?
We have developed DVG assistance solutions for your digital health products to help you get through the assessment process. If you want more details, please book a call with us.








