Imagine if doctors could prescribe your app like they do drugs. How would it transform your business model if you could be paid directly by a patient's health insurer? Welcome to the world of digital therapeutics.
Digital therapeutics is the next big opportunity in digital health. Digital health apps can now be prescribed by doctors and paid for by insurers. Suddenly, there's a new business model for your application. However, to benefit from this, you will need to prove the security, quality and effectiveness of your app. Read on to find our how Chino.io can help you be at the forefront of this revolution.
Background: What is digital therapeutics?
Digital therapeutics is the catchall for digital health applications that actively support and treat patients. These apps are another weapon in a doctor’s arsenal of treatments he can prescribe. Examples include:
- Virtual reality apps to help psychiatric patients
- Coaching apps that treat drug addictions
- Apps that help people with diabetes to stabilise their blood sugar
But there are countless other examples. What distinguishes digital therapeutics from other digital health applications is that they have a demonstrable impact on patient outcomes.
What does this mean for my application?
Up to now, doctors have relied on drugs and therapies to treat patients. Now they will be able to prescribe applications and recover the money from insurers. Governments are waking up to the opportunities this offers for improving public health and saving money. As a result, new laws like the German Digitale-Versorgung-Gesetz (DVG) are being proposed. Similar things are already happening with the UK's NHS, the FDA in the US and in other countries.
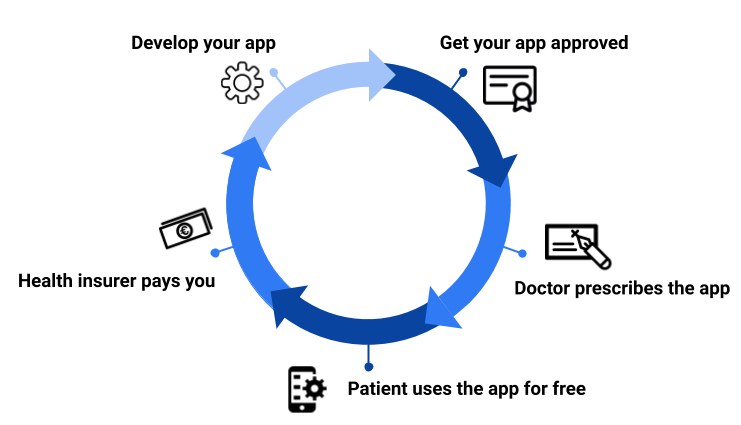
What does this mean for my business model?
Quite simply, it opens up a new business model for any companies who develop digital health applications. The drugs industry is worth over a trillion dollars globally. If just a fraction of that is diverted to digital therapeutics, that will be transformative. Startups will find it easier to raise capital, established companies will risk bigger investments and new companies will enter the digital health market. Of course, this will also generate increased competition and growth in this already growing sector. This means only companies with the most effective apps will survive. It is important to note that the price you can charge users for your app will need to be negotiated with the appropriate authority, e.g. the GKZ-Spitzenverband in Germany and the NHS in the UK.
What do I need to do next?
As a developer, you need to take steps to ensure the quality, effectiveness and security of your applications. This includes being assessed for compliance with regulations like GDPR and MDR, implementing suitable quality management processes and ensuring all health data is protected and secured. Here is a brief summary:
Data protection and security: Both the DVG and the NHS assessments require you to comply with GDPR and data security standards. For health applications, this means you need significant investment in technology.
Quality management: You need to prove that your app meets the quality requirements for healthcare software. This probably means getting certified under the new MDR and undertaking ISO 13485 certification.
Medical effectiveness: The German DVG allows your apps to be funded for one year while you collect data that proves their effectiveness. After that, if you can't convince BfArM that you app is effective, it will no longer be funded. Similar rules apply in other jurisdictions.
None of these requirements are actually new. But up until now, developers have usually been able to self-certify their compliance. That was already changing with MDR, but this will accelerate that change.
How can Chino.io help you?
We are experts in all the data security and legal compliance requirements for healthcare. Indeed, we are actively using our expertise to help shape the formal requirements for DVG. As this represents such a huge opportunity for the digital health space, we are offering you a chance of a 30-minute call to discuss exactly how it will impact your company. Click below to schedule a call.
Get a free expert assessment